Zf-work-pharma for Manufacturers
Your company manufactures pharmaceutical products that are subject to serialization requirements, and you need to implement serialization in an environment with a wide variety of customer systems and established packaging infrastructure.
Then we offer the best solution for implementing your local serialization processes:
Our Level 3 track-and-trace solution Zf-work-pharma and our expertise in serialization processes and integration with third-party systems.
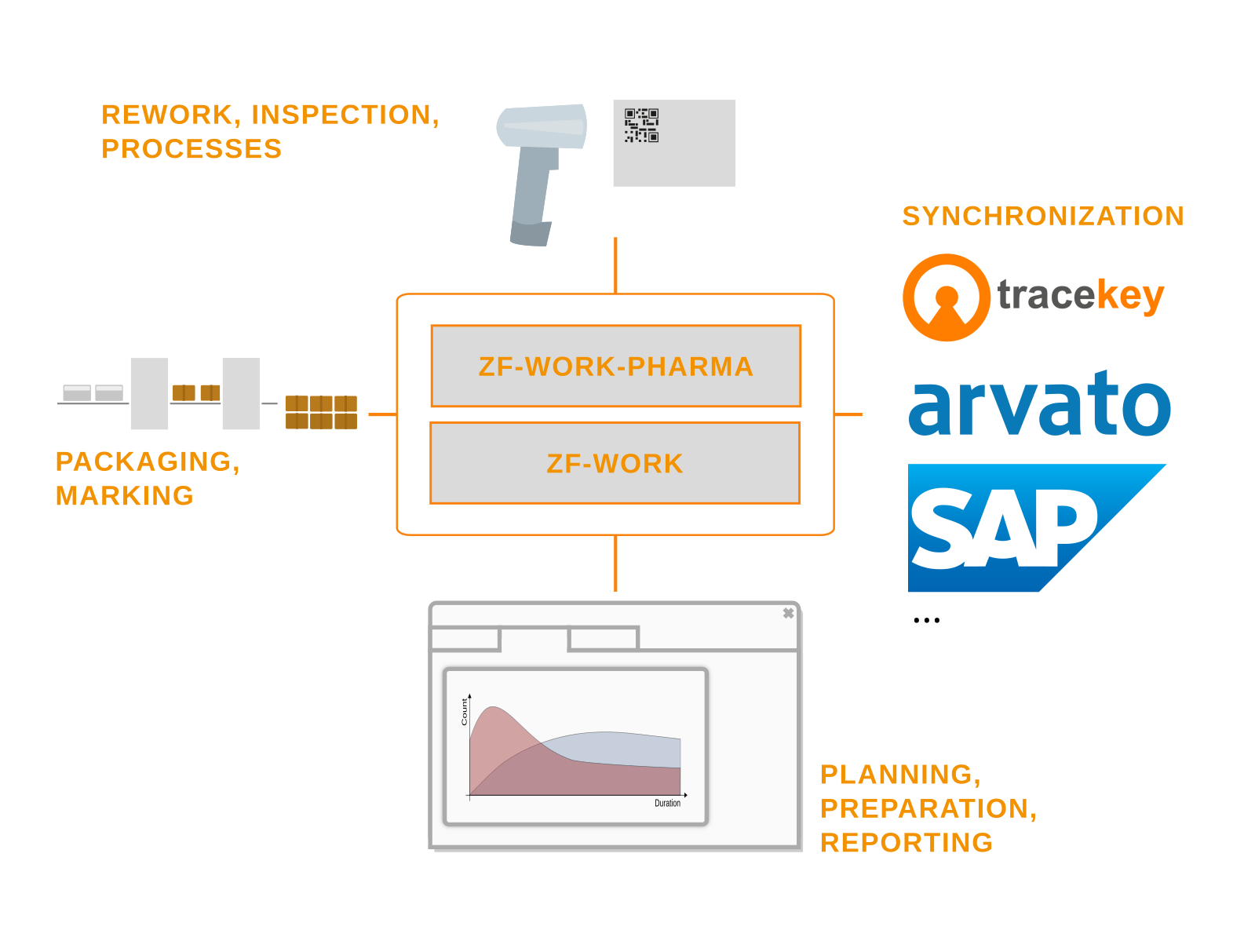
On-site control of serialization and integration with new and old packaging and inspection systems.
Production preparation and coordination with the packaging line is the core business of zf-work-pharma.
Production orders can be moved between machines, designated for commercial samples, or designed to coordinate the pre-printing of collapsible cartons.
We also do not shy away from connecting exotic packaging systems.

Order preparation and serial number requests with tracekey, Arvato, Tracelink, and others.
Zf-work-pharma supports the connection of standard Level 4 systems and, on request, non-standard Level 4 systems as well.
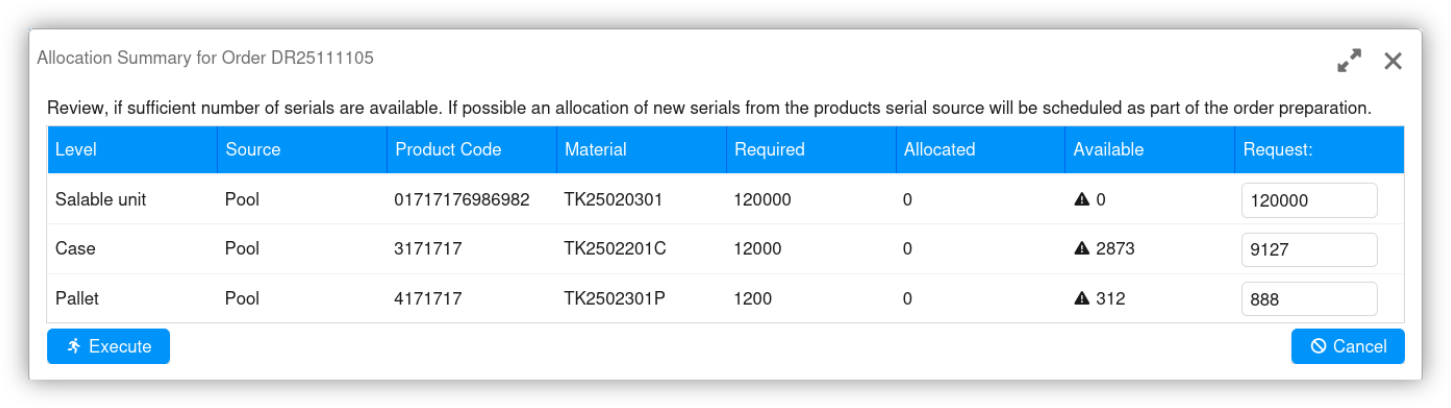
Master data-based configuration simplifies serial number management, from serial number allocation to feedback.
Support for regional requirements
Zf-work-pharma supports master data-based configuration of GS1 serialization requirements and regional specifications, such as the application of national reimbursement numbers or Russian crypto codes.

Support for aggregation, bulk changes, rework

Zf-work-pharma supports aggregation scenarios in production and rework. If something goes wrong, bulk changes via table upload enable simple and traceable repairs.
Initial and update reporting for batches and post-processing
Zf-work-pharma is based on a genuine track & trace core and can not only generate batch reports, but also record genuine delta reporting, including aggregation changes.

Customizable with validatable software logistics
Based on the zf-work application platform, existing standard processes can be expanded, supplemented, modified, and integrated with technical interfaces of your local systems. Our distributed software logistics make upgrades, maintenance, and customization simple, transparent, and validatable.

If something is missing or a special case needs to be resolved, the code and documentation remain with you, with maximum transparency and fairness.
